- Mechanical and microstructural properties of synthetic fiber reinforced geopolymers exposed to acidic environments and freeze-thaw cycles in seawater
Zahraa Mohammed Kamil Al-Mayali and Mahmoud Ziada*
Department of Civil Engineering, Istanbul Aydin University, Istanbul, Türkiye
This article is an open access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
This study investigated the durability performance of fresh and hardened geopolymer mortars with and without synthetic fibers. To examine two separate durability tests, one group of samples was first exposed to acid exposure, and the other group was subjected to freeze-thaw cycles in seawater. Additionally, a third group was left unexposed for comparison. Fiber-containing and fiber-free geopolymer samples were examined for their fresh and hardened properties before and after durability testing. Physical, mechanical, and microstructural analyses were performed for this purpose. The effect of fiber on the geopolymer samples differed before and after durability testing. The highest pre-durability compressive strength was obtained from the geopolymer sample with 1.5% fiber, recording a 21.62% increase compared to the samples without fiber. On the other hand, after acid exposure and freeze-thaw cycles in seawater, the samples with 1.5% fiber had the lowest compressive strengths, with reductions of 24.07% and 11.51%, respectively, compared to the fiber-free samples. Thus, this study emphasizes that the effect of fiber in geopolymer mortar samples may show differences before and after durability exposures.
Keywords: Metakaolin, Macro synthetic fiber, Freeze-thaw, Sulfuric acid.
Geopolymers offer a more sustainable composite material compared to cementitious composites [1]. Many of the binder materials used in geopolymers can be obtained from industrial waste [2]. Furthermore, previous studies have demonstrated that geopolymer composites can be produced as high-early-strength composites [3, 4]. Geopolymers have also demonstrated significant durability. This has led to the usability of geopolymers in many types of structures [5, 6]. It should be emphasized that the properties of geopolymer composites differ depending on the binder materials used in geopolymers. Previous work utilized metakaolin and fly ash as binder materials, resulting in metakaolin achieving higher early strength in geopolymers compared to fly ash [7, 8].
The insufficient acid-resistant properties of cementitious composites can lead to damage and collapse in cement-based structures exposed to acidic environments. An acid penetration-preventing layer can be created on the composite to reduce the risk of acid attack [9]. However, layering has several disadvantages, including high costs and application difficulties [10]. Geopolymers can be resistant to acid depending on the binder material used. In a study by Davidovits et al. [11], a metakaolin-based geopolymer was produced, and a low level of acid exposure was observed in the resulting geopolymer. Another research group [12] highlighted that increasing metakaolin in geopolymer increased acid resistance. Another perspective [13] states that not only the binder but also the molarity of the activator plays a significant role in acid resistance.
Another important durability effect is the freeze-thaw cycle to which buildings are subjected in cold weather. The freeze-thaw cycle of cementitious composites can cause serious damage [14]. This damage occurs due to the freezing and expansion of water in the concrete voids [15]. A previous study had used nanomaterials to increase freeze-thaw resistance [16]. The freeze-thaw effects on geopolymers containing different binders have been investigated [17–19]. However, the amount of research investigating the impact of freeze-thaw on the properties of geopolymer composites with fiber is somewhat restricted [20, 21]. Aygörmez et al. [21] examined the impact of freeze–thaw impact on the microstructural and mechanical properties of geopolymer. They reported a decline in flexural strength and compressive strength.
The use of fibers to enhance the performance of geopolymers, similar to that in cementitious composites, has gained importance. In general, the expected property of fiber is to prevent or limit the development of cracks [22, 23]. Furthermore, fibers play a significant role in increasing the flexural and tensile strength of different composites [24]. The superior performance of fibers is attributed to their elastic modulus and strength, which have helped to popularize their use [25, 26]. These properties contribute to the enhancement of ductility in fibrous geopolymer composites. Therefore, geopolymers are relatively brittle materials, and the use of fibers to increase their flexural and tensile strength has been encouraged [27]. Previous studies [28, 29] have found that geopolymer samples containing basalt fibers have high mechanical properties. Furthermore, another study found that the addition of steel fibers significantly increased the flexural strength of geopolymers [30].
Although prior research has investigated the durability of geopolymers in acidic environments or under freeze–thaw conditions, there exists a notable deficit in research on the effects of freeze–thaw cycles in seawater. Addressing this deficiency is essential since comprehending the behavior of geopolymers in these settings might provide significant insights into their use in cold places subjected to marine environments. This renders the current work especially pertinent for the development of resilient geopolymer materials appropriate for severe coastal ecosystems.
Substances
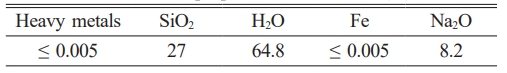
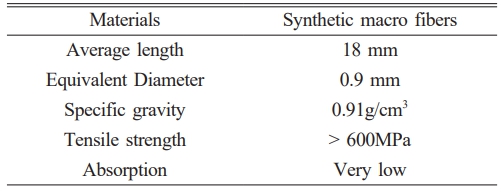
In this work, sodium hydroxide (NaOH) and sodium silicate (Na2SiO3) were used as catalysts and activators in geopolymer mortars. Tables 1 and 2 show their chemical compositions. Slag and metakaolin were used as binders. Table 3 shows their chemical compositions. The table shows that metakaolin contains 40.23% Al2O3 and 56.1% SiO2. The substantial quantity of silicon and aluminum oxide facilitates the production of silicon-oxygen-aluminum tetrahedra inside the geopolymer matrix [31]. Slag has 35.58% CaO, which facilitates water absorption, improving the density of the geopolymer matrix and its suitability for frost resistance and strength [32]. Macro synthetic fibers were used as reinforcing materials for geopolymer mortars. Sulfuric acid served as an acidic solution to evaluate resistance to acid. Fig. 1 shows the materials used to make geopolymer mortars.
Fabrication and proportions of geopolymer specimens
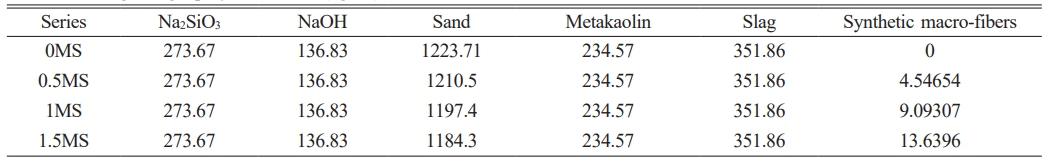
Prior to preparing the geopolymer mortar used in this study, a 12 M sodium hydroxide mixture was created by incorporating 240 g of sodium hydroxide into a 500 ml volumetric flask and filling the flask to the 500 ml mark. The solution was allowed to cool at 20 °C for one day and then mixed with a sodium silicate solution. Synthetic macro fibers were then added to the sand at ratios of 0, 0.5, 1, and 1.5% and mixed together. Binders (slag and metakaolin) were then added to the sand-fiber mixture and mixed for two minutes. The activated solution was then added to the dry mixture and mixed for two minutes using a mixing device. The mixtures were then placed in prepared-lubricated 50 mm × 50 mm × 50 mm and 40 mm × 40 mm × 160 mm molds for 24 hours. The samples were subsequently situated on a vibrating platform for one minute at 50 Hz to reduce entrapped air. After one day, Fig. 2 shows how the materials were mixed and poured into the molds. The samples were removed from the molds and stored in an oven at 23 °C for 28 days. Table 5 shows the quantities of the mixtures used in this study.
Performed tests
This study evaluated slump flow values, ultrasonic pulse velocity (UPV), water absorption, compressive strength, flexural strength, and mass change to assess the effect of fiber addition to geopolymer mortar on the resistance to sulfuric acid, freeze-thaw conditions, and mechanical properties of slag-based geopolymer mortar. In addition, scanning electron microscope (SEM) analysis was performed to examine the microstructural characteristics of the samples, and X-ray diffraction (XRD) was conducted to ascertain the crystalline components of the specimens. A mini slump experiment was conducted for the mixes containing varying fiber ratios to evaluate the consistency level of the fresh blends. The fresh blend was positioned inside a copper mold on 2 levels, and each level was tapped with a wooden-copper hammer. The conical mold was then lifted, and the surface was manually struck 25 times. The slump flow values in both directions were evaluated and averaged. Fig. 3 shows the mini slump experiment of geopolymer samples.
Compressive strength testing was performed on 50× 50×50 mm cubic specimens using a 250 kN compression machine and a loading rate of 0.6 MPa/s according to ASTM C109 [33]. Flexural strength testing was performed on 40×40×160 mm specimens at a loading rate of 0.469 MPa/s to determine the flexural strength according to ASTM C348 [34]. These tests were first performed on the geopolymer specimens before placing them in acid and before the freeze-thaw in seawater experiment. Figure 4 shows the compression and flexural experiments. In addition, a capillary water absorption test was performed on the geopolymer specimens according to ASTM C1585 [35]. The prismatic specimens were desiccated in a heated oven at 105 °C for one day. Subsequently, their weights were measured, and their dry weight was taken. Vaseline was then applied to all surfaces of the specimens, except for the surfaces in contact with water. Following that, the specimens were submerged in 5 mm of water, and the samples were extracted at different times, and the weight of the immersed specimens (Ww) was provided.
Additionally, sulfuric acid was diluted to a 5% concentration, and the specimens were soaked in a 5% solution of acid at 20 °C for twenty-eight days, as shown in Figure 5. The solution was renewed every 7 days to maintain a constant concentration throughout the exposure period. The compressive strength of the samples was retested at 14 and 28 days, and similarly, a flexural strength experiment was performed on the geopolymer specimens after 14 and 28 days of acid treatment. Additionally, a weight change test was performed before and after 14 and 28 days, and a UPV test was performed on cubic samples before and subsequent to 14 and 28 days of the treatment of acid according to ASTM C597 [36] in Fig. 6.
A freeze-thaw test in seawater was also performed on the geopolymer samples with and without fiber. Initially, the seawater-saturated samples were placed in a freezer for 12 hours at -20 °C. Then, the samples were taken and immersed in seawater for 12 hours at +20 °C to constitute one freeze-thaw cycle in seawater. Thus, each cycle was equalized by one day, and this test was continued for 28 days. During this period, compressive and flexural strength tests were performed on the 14th and 28th days. Also, weight change tests were performed on the 14th and 28th days of the test. UPV testing of cubic samples was conducted before and after 14th and 28th days of testing in accordance with ASTM C597 [36]. Fig. 7 shows the samples during the freezing and thawing stage.

|
Fig. 1 Materials used to make a fiber-reinforced slag-metakaolin geopolymer mixture: (a) Slag, (b) Metakaolin, (c) Synthetic macrofibers, (d) Sodium hydroxide, and (e) Sodium silicate. |

|
Fig. 2 Steps for mixing geopolymer mortar: (a) Combining activators, (b) Blending sand and fiber, (c) Conducting a dry mix of all ingredients, (d) Mixing activators with the dry mix, and (e) Molding the mortars. |

|
Fig. 3 Slump flow experiment of slag-metakaolin-based geopolymer samples reinforced with fiber. |

|
Fig. 4 Testing of geopolymer mortar specimens with and without fiber after 28 days of casting: (a) Compressive, and (b) Flexural strength. |

|
Fig. 5 Process of placing synthetic macro-fibers -reinforced slag-metakaolin-based geopolymer samples into sulfuric acid. |

|
Fig. 6 UPV testing of geopolymer samples after 28 days of durability. |

|
Fig. 7 A freeze-thaw cycle of synthetic macro fibers-reinforced slag-metakaolin-based geopolymer samples: (a) after freezing at -20 °C, and (b) after thawing at +20 °C in seawater. |
Slump flow values
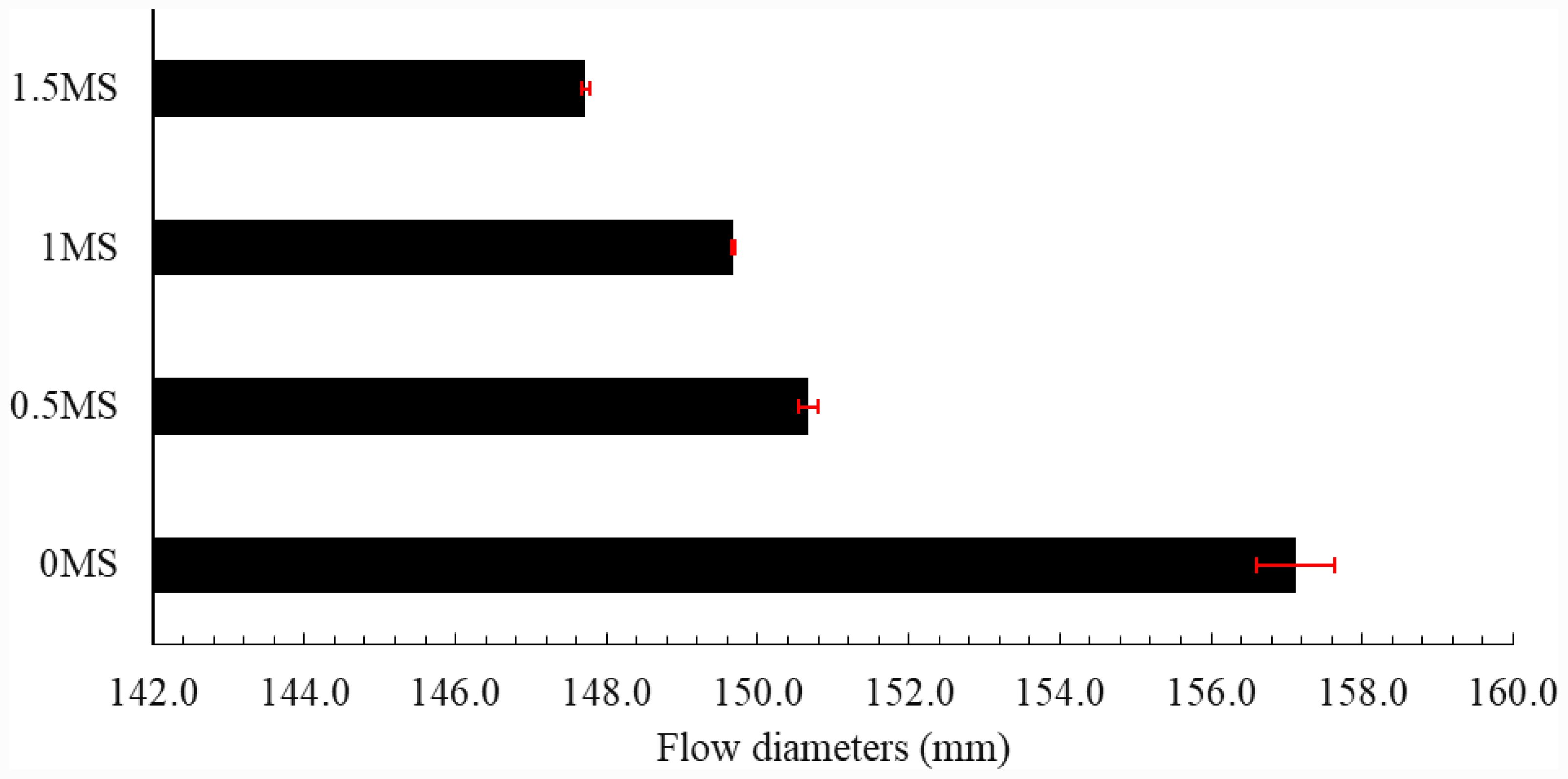
A slump flow test was conducted to evaluate the workability of geopolymer samples. This test provides accurate performance assessments and, despite its inherent limitations, is widely used to ensure the stability of blends and to guarantee desired composite specifications. The development of stable blends with low slump rates has contributed to the increased adoption of this test as a key tool for evaluating and improving material properties. Figure 8 shows the average flow rates for fiber-reinforced geopolymer samples. The highest flow rate of 157.1 was achieved for the 0MS geopolymer sample. Increasing the fiber content in the geopolymer samples resulted in lower flow values, with the diameter decreases of 4.09%, 4.73%, and 5.97% for the 0.5MS, 1MS, and 1.5MS samples, respectively, in comparison to the 0MS. The augmentation of synthetic fibers diminishes the slump flow of geopolymer mixtures, since the fibers impede the mobility of the fresh mortar by augmenting internal friction and decreasing workability. Several studies have reported similar observations [26, 30, 37].
Curves and coefficients of capillary water absorption
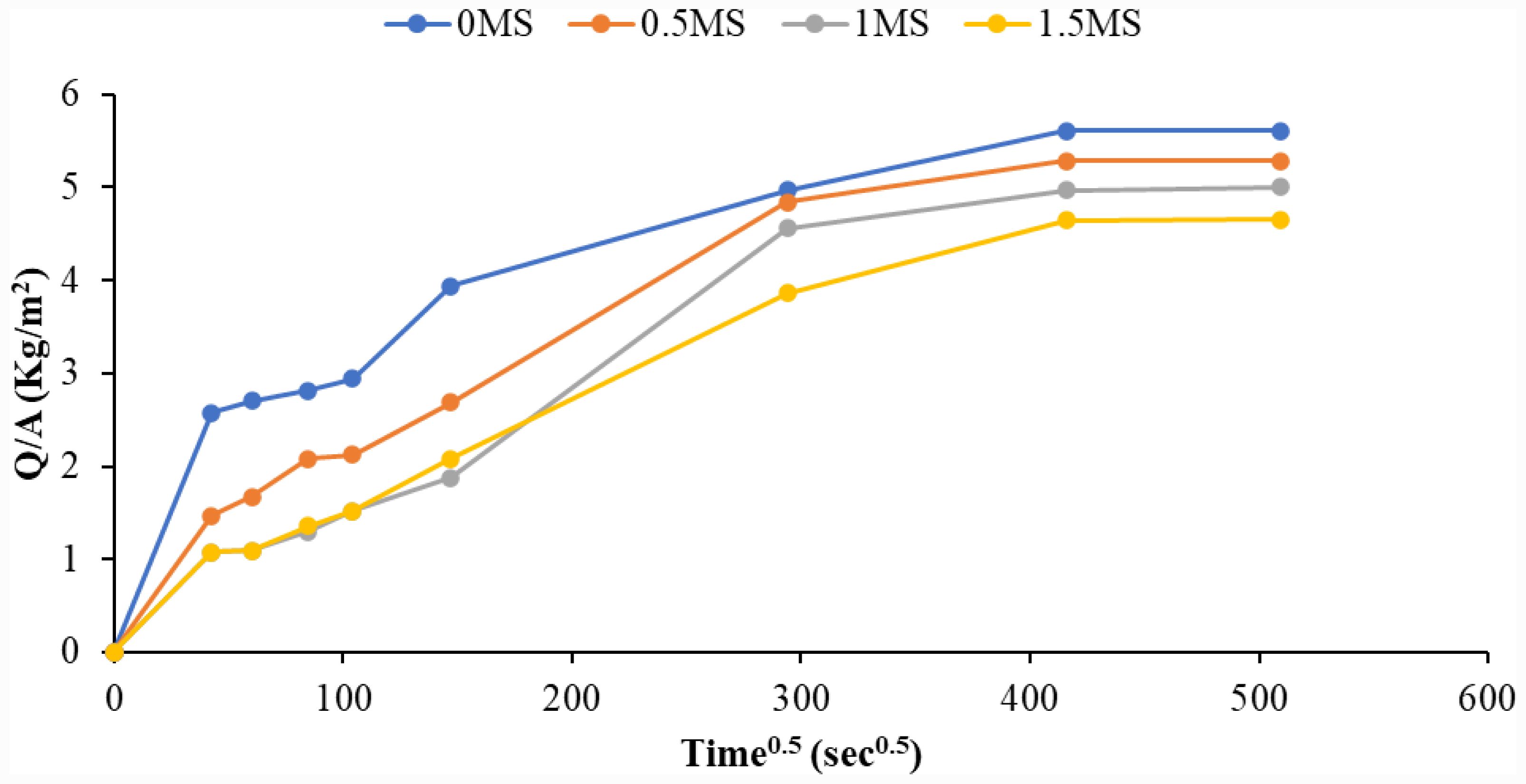
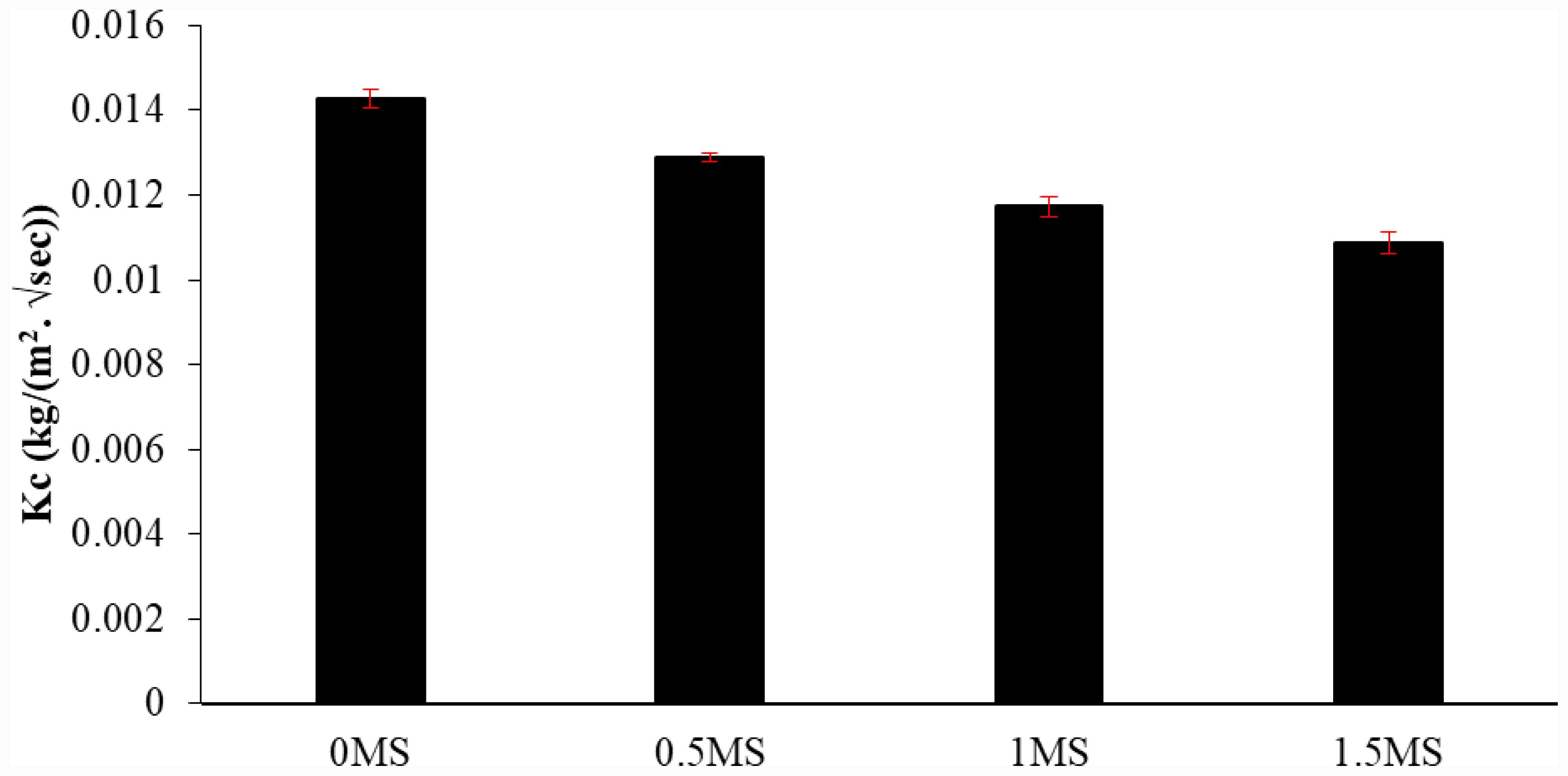
Water absorption curves and coefficients were found to evaluate the density and permeability of hardened geopolymer specimens. Fig. 9 shows the curves for geopolymer mortar specimens using different synthetic macro fiber contents. The results revealed that as the synthetic macro-fiber content increased, the water absorption capacity of the geopolymer samples decreased. The sample containing 1.5MS exhibited the smallest value in comparison with the remaining specimens. The capillary coefficient of absorption (kc) was computed for a more precise comparison, as shown in Fig. 10. It was noted that the capillary absorption coefficient decreased with increasing synthetic macro fiber content within the geopolymer samples compared to 0MS. The percentage decrease in the kc coefficient compared to 0MS was 9.69%, 17.85%, and 23.85% for 0.5MS, 1MS, and 1.5MS, respectively. This can be interpreted as the random dispersion of fine and large fibers, allowing for the suture of microcracks and preventing the emergence of new cracks in the geopolymer matrix. This mechanism translates into an elevated density of the composition, thus reducing the capacity for absorbing water [38]. In addition, due to the hydrophobic nature of the synthetic macro-fiber, samples containing synthetic macro-fiber absorbed less water than those devoid of fibers [26].
Compressive strength
Compressive strength of specimens exposed to sulfuric acid
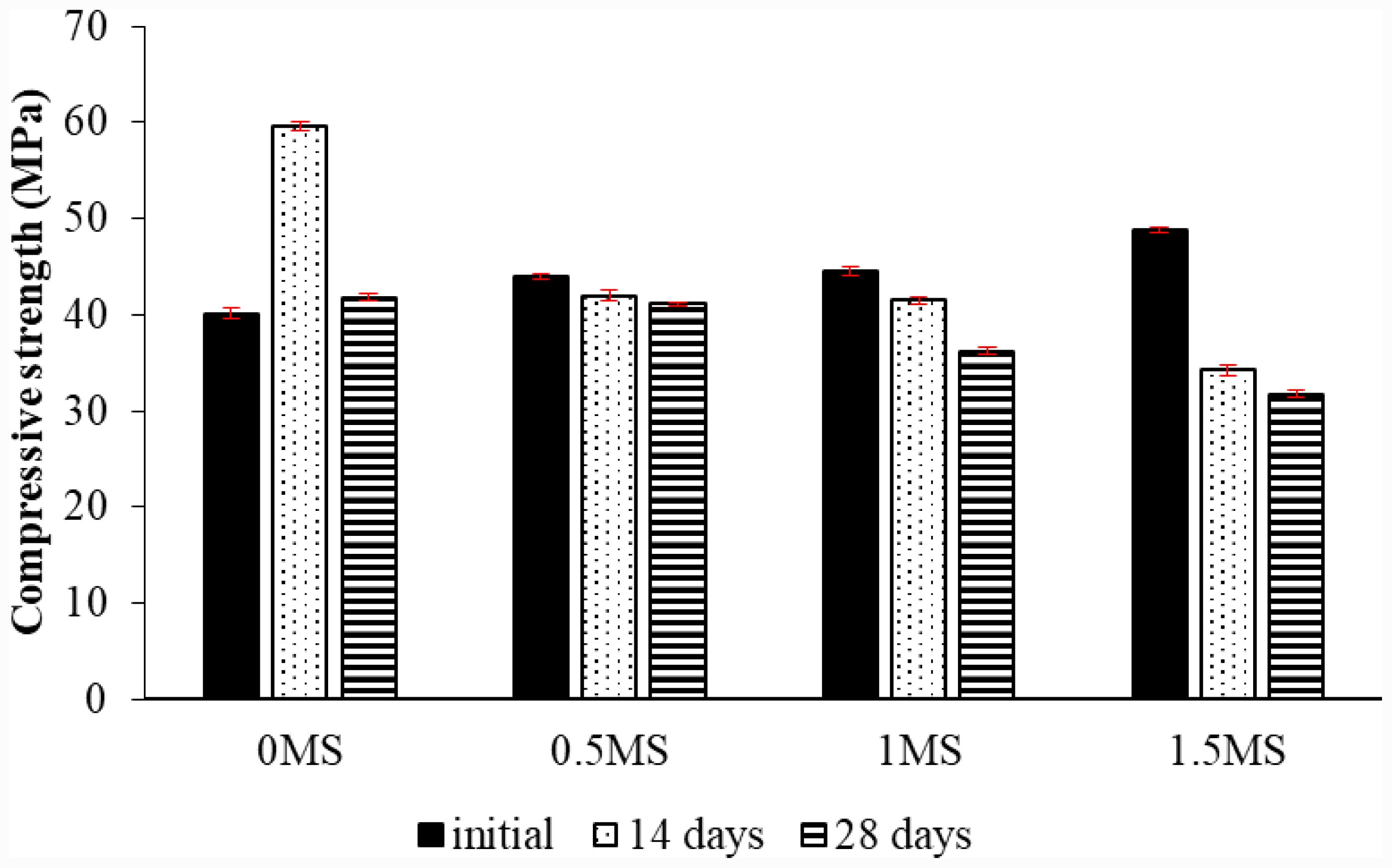
A compressive strength test was conducted to determine the mechanical properties of slag-metakaolin-based geopolymer samples with different fiber contents. A group of samples was underwent exposure to sulfuric acid for 14 and 28 days. Three samples were examined for each fiber content before and after 14 and 28 days of acid treatment. The average values were then found. Fig. 11 depicts the compressive strength values of the specimens with differing fiber content prior to and subsequent to fourteen and twenty-eight days of acid exposure. The results showed that the slag-metakaolin geopolymer samples with a ratio of 1.5MS before exposure to acid achieved the highest compressive strength compared to samples with ratios of 0MS, 0.5MS, and 1MS. The integration of synthetic fibers improves the compressive strength of geopolymer mixtures by connecting microcracks and postponing their advancement under stress. Furthermore, the fibers enhance stress distribution within the matrix, resulting in a more compact and resilient composite structure [39].
The geopolymer sample with a ratio of 1.5MS achieved the lowest compressive strength after 28 days of exposure to acid, compared to samples with lower ratios. The percentage decrease in compressive strength for samples 0.5MS, 1MS, and 1.5 after 28 days of exposure to acid was 1.62%, 13.37% and 24.07% respectively, compared to samples with 0MS. In addition, the mortars after being immersed in the acidic solution for two weeks achieved valuable results, as the compressive strength of the geopolymer sample with the content of 0MS increased by 48.47% compared to before exposure to acid. This is due to the fact that these samples do not contain fibers and their internal structure consists of metakaolin and slag, where the reactive aluminosilicate properties of metakaolin form a three-dimensional geopolymer gel network, which enhances the density, strength, and mechanical properties [12, 40].
While samples with ratios of 0.5MS, 1MS, and 1.5MS gave lower compressive strength after 14 days of exposure to acid, the percentage decrease for the slag-metakaolin-based geopolymer samples with contents of 0.5MS, 1MS, and 1.5MS compared to before exposure to sulfuric acid was 4.49%, 6.74% and 29.03%, respectively. This may be attributed to the deterioration of the contact area between the fibers and the geopolymer structure due to the effect of the acid, which leads to acid penetration along the fibers, increased microcracks around the fibers, and loss of bonding, which leads to loss of the fibers’ contribution to compressive strength. This was explained by M. Ohno and V.C. Li in their study on the effect of the presence of fibers in samples exposed to acid [41]. After 28 days of exposure to the acid, the compressive strength of 0MS also increased compared to its strength before the acid, where the percentage of increase was 4.17%, but it decreased compared to its strength at 14 days of exposure to the acid. This is attributed to the fact that in cement, concrete, and geopolymers, it is believed that prolonged exposure to sulfuric acid causes deterioration in the microstructure of geopolymer composite materials [42, 43]. In addition, the compressive strength of 0.5MS, 1MS, and 1.5MS slag-metakaolin-based geopolymer samples decreased after being immersed in acid for 28 days, in contrast to their strength prior to and following the acid immersion. The values of compressive strength diminish from 14 to 28 days of acid exposure due to the extended contact with acidic media, which promotes the leaching of alkaline ions and the dissolution of reaction products, resulting in slow matrix degradation and a weakening of the binding phases [12].
Compression test of geopolymers tested by freeze-thaw cycle in seawater
In the second part of the experiment, samples were exposed to freeze-thaw conditions for 14 and 28 days. Three samples for each fiber content were tested before and after the 14-day and 28-day freeze-thaw experiment. The average values were then calculated as illustrated in Fig. 12. The results showed that the 1.5MS component geopolymer sample yielded the lowest compressive strength after 28 days of freeze-thaw cycling compared to samples with lower fiber contents. The percentage decrease in compressive strength of the 0.5MS, 1MS, and 1.5MS samples after 28 days of freezing and thawing was 5.25%, 8%, and 11.50%, respectively, compared to the 0MS. Additionally, geopolymer samples after 14 days of freezing and thawing yielded mixed results, with the compressive strength of the 0MS content geopolymer sample increasing by 5.37% compared to before freezing and thawing. This is due to the fact that these samples do not contain fibers, and their internal composition consists of metakaolin and slag. The existence of elevated concentrations of active compounds with alkaline activators promotes rapid dissolution of the silicoaluminate components [44]. As a result, hydration and geopolymer reactions are accelerated and stimulated when immersed in water, resulting in the creation of a polymer gel that occupies the gaps inside the substance, thereby markedly improving its strength and density [45]. Thus, this reaction plays an effective role in contributing to the initial high compressive strength. In addition, the compressive strength of geopolymer is enhanced by exposure to freeze-thaw cycles in seawater, as dissolved salts, particularly K⁺ ions, infiltrate and precipitate inside the pore structure during freezing and thawing, therefore plugging microcracks and purifying the matrix. K⁺ ions may engage in further geopolymeric processes, facilitating further gel formation that densifies the matrix and improves mechanical performance [46, 47].
While samples with ratios of 0.5MS,1MS, and 1.5MS exhibited lower compressive strength after 14 days of freeze-thaw, the percentage decrease in the slag-based and metakaolin-based geopolymer samples with contents of 0.5MS, 1MS, and 1.5MS compared to before the freeze-thaw experiment was 9.06%, 12.77%, and 25.40%, respectively. The compressive strength diminishes as the amount of fiber increases. A similar tendency was seen in a previous study [48]. The inclusion of fibers disrupts the adhesion of the binding components, rendering the substrate prone to fracturing. The 0MS samples’ compressive strength increased following 28 days of freeze-thaw compared to their strength before the experiment, where the percentage increase was 0.88%. However, it decreased compared to its strength at 14 days of experimental exposure. This is attributed to the fact that the reactions slow down and are completed when the substance nears its ultimate configuration and characteristics. The compressive strength of the slag- and metakaolin-based geopolymer samples decreased after 28 days of freezing and thawing in seawater compared to their strength before and after 14 days of the experiment. The compressive strength of geopolymer diminishes from 14 to 28 days of freeze-thaw exposure due to repeated cycles of freezing and thawing that promote micro-cracking, enhance pore connectivity, and progressively compromise the matrix structure [49].
Flexural strength
Flexural strength test of geopolymer mortars treated with sulfuric acid.
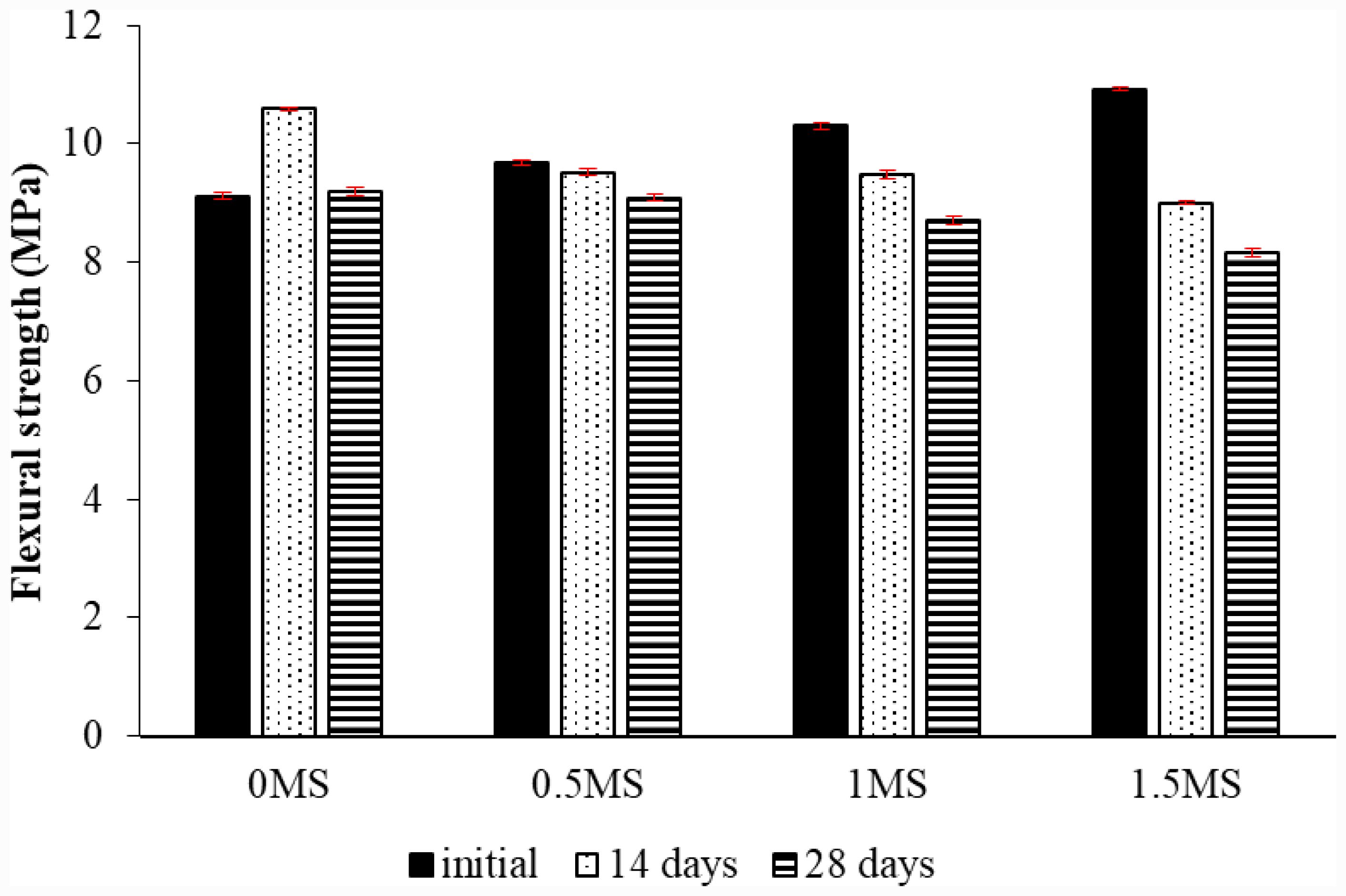
A flexural test was conducted to determine the mechanical properties of slag- and metakaolin-based geopolymer samples with different fiber contents. The experiment was conducted in two parts. In the first part, a set of samples underwent exposure to sulfuric acid for 14 and 28 days. Three samples for each fiber content were examined after and before being subjected to the acid effect for 28 and 14 days. The average values were then found. Figure 13 illustrates the bending strength values of the specimens without and with fiber contents prior to and subsequent to fourteen and twenty-eight days of exposure to acid. The results showed that the slag-metakaolin geopolymer samples with a 1.5MS ratio before acid exposure exhibited the highest flexural strength compared to samples with 0MS, 0.5MS, and 1MS ratios. The use of synthetic fibers enhances the flexural strength of geopolymer composites by bridging emerging fractures and improving load distribution throughout the matrix. This reinforcing method inhibits fracture propagation and enhances toughness, yielding a more ductile and robust material [50].
Meanwhile, the 1.5MS geopolymer exhibited the lowest flexural strength after 28 days of acid exposure compared to samples with lower ratios. The percentage decrease in flexural strength of the 0.5MS, 1MS, and 1.5MS samples after 28 days of acid exposure was 1.21%, 5.43%, and 11.30%, respectively, compared to the 0MS samples. Furthermore, geopolymer samples after 14 days of acid exposure showed mixed results, with the 0MS geopolymer sample showing a 16.21% increase in flexural strength compared to before acid exposure. While the 0.5MS, 1MS, and 1.5MS samples showed lower flexural strength after 14 days of acid exposure, the percentage decrease for the slag- and metakaolin-based geopolymer samples with 0.5MS, 1MS, and 1.5MS contents after 14 days compared to before sulfuric acid exposure was 1.76%, 7.89%, and 17.51%, respectively. This may be attributed to the deterioration of the contact area between the fibers and the geopolymer structure due to the acid effect, which leads to acid penetration along the fibers, increased microcracks around the fibers, and loss of bonding, resulting in a loss of fiber contribution to bending strength [41]. The strength of the 0MS specimens also increased subsequent to twenty-eight days of acid effect compared to their strength before the acid, where the increase was 0.86%, but it decreased in comparison to their strength following being subjected to the acid effect. This is attributed to the fact that in cement, concrete, and geopolymers, prolonged exposure to sulfuric acid is believed to cause microstructure deterioration of geopolymer composite materials [43]. The flexural strength of the geopolymer specimens declined following 28 days of acid exposure in comparison to their strength following 14 days of acid exposure [51].
Flexural strength test of geopolymer specimens subjected to cycles of freezing and thawing
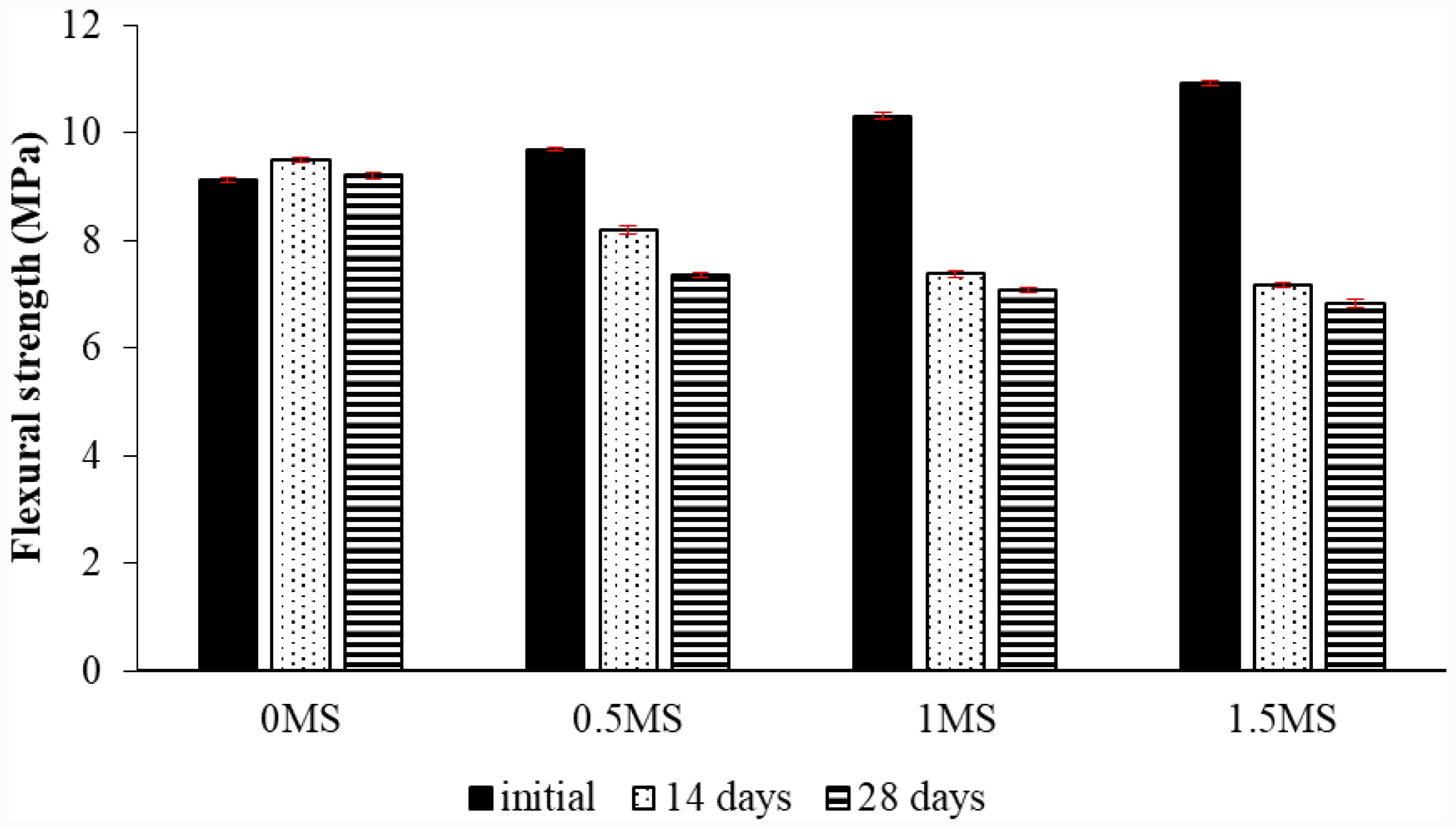
The specimens were exposed to freeze-thaw conditions for 14 and 28 days in the second part of the experiment. Three samples for each fiber content were tested before and after the 14-day and 28-day freeze-thaw experiment. The average values were then obtained. Fig. 14 illustrates the bending strength findings of the geopolymer mortars without and with fiber contents, subsequent to and before the 14 and 28-day freeze-thaw experiment. The results showed that the 1.5 MS geopolymer sample achieved the lowest flexural strength after 28 days of freeze-thaw compared to samples with lower fiber contents. The percentage decrease in flexural strength of the 0.5MS, 1MS, and 1.5MS samples after 28 days of freeze-thaw was 20.21%, 23.01%, and 25.84%, respectively, compared to 0MS. Furthermore, geopolymer samples after 14 days of freeze-thaw yielded different results, with the bending strength of the geopolymer mortar containing 0MS increasing by 4.26% in comparison to that before freeze-thaw. The flexural strength of geopolymer subjected to freeze–thaw cycles in seawater improves owing to the establishment of a stable network that bolsters resistance to freeze–thaw deterioration and chemical corrosion [52]. While the 0.5MS, 1MS, and 1.5MS samples exhibited lower flexural strength after 14 days of freeze-thaw, the percentage decreases of the slag-metakaolin-based geopolymer samples with 0.5MS, 1MS, and 1.5MS contents compared to that before freeze-thaw were 15.44%, 28.37%, and 34.34%, respectively. The flexural strength of geopolymer samples with synthetic fibers may diminish during freeze-thaw conditions, since repeated cycles of freezing and thawing facilitate fiber pull-out and micro-cracking at the fiber-matrix interface, therefore compromising load transfer efficacy. When fiber content or distribution is inadequate, these stress concentrations serve as initiating sites for structural deterioration, resulting in diminished overall flexural performance [53]. Flexural strength dropped
as fiber content climbed. The incorporation of fibers interferes with the adherence of the components, rendering the substrate vulnerable to breaking due to moisture penetration into the contact area between the fibers and the geopolymer structure. The flexural strength of the 0MS sample increased after 28 days of freezing and thawing compared to its strength before the experiment, with a percentage increase of 0.97%. However, it decreased compared to its strength at 14 days of experimental exposure. The flexural strength of the slag- and metakaolin-based geopolymer samples decreased by 0.5 MS, 1 MS, and 1.5 MS after 28 days of freezing and thawing, compared to their strengths before and after 14 days of the experiment.
UPV findings
UPV of geopolymer mortars after sulfuric acid.
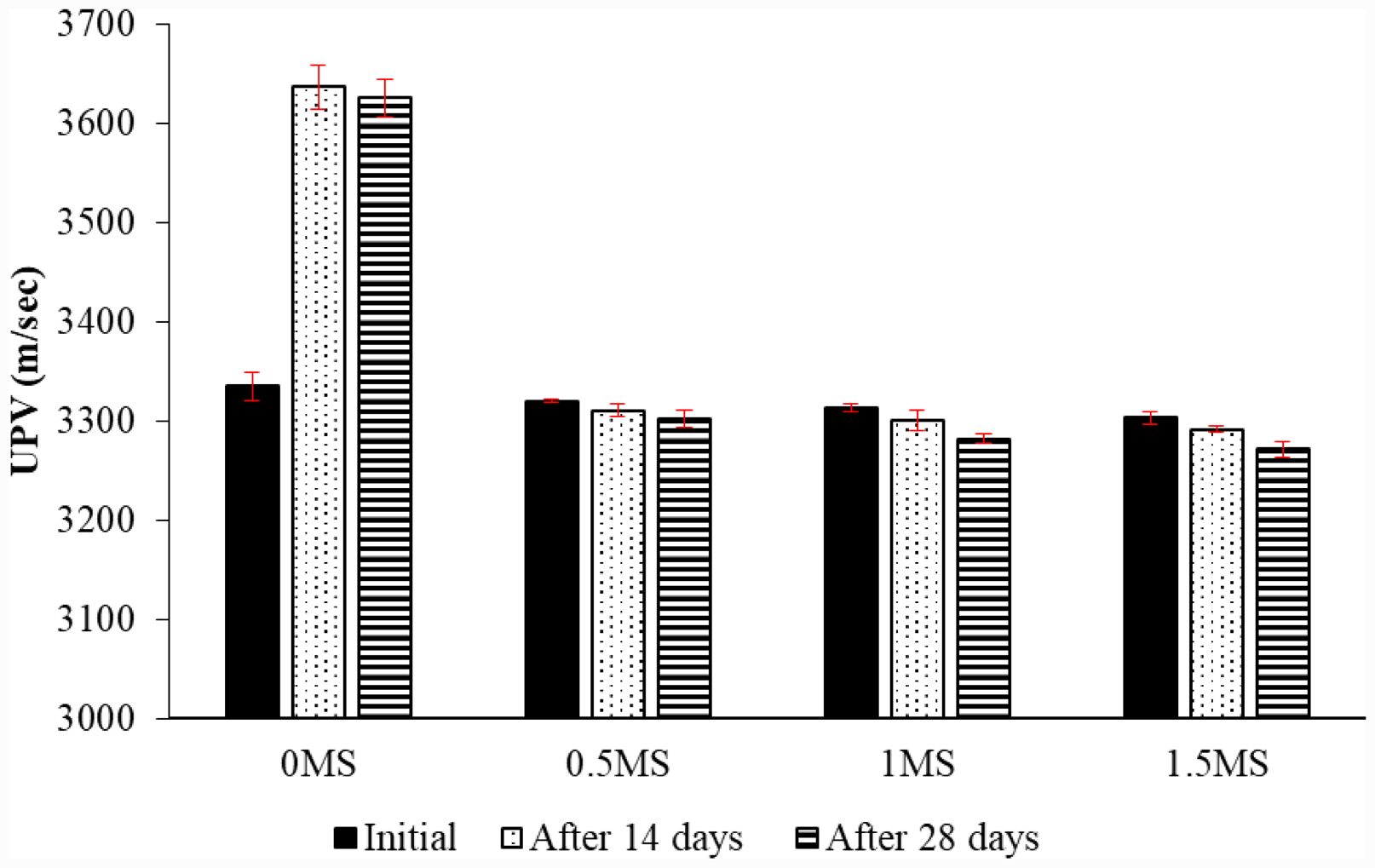
Figure 15 shows the UPV values of geopolymer samples exposed to acid treatment. UPV values were obtained after immersion of the samples in sulfuric acid solution for 14 and 28 days and compared with control values. Furthermore, this experiment was also used to investigate the effect of fiber on the geopolymer matrix. Among the fibrous and non-fibrous geopolymer samples exposed to acid, the highest UPV value was obtained from the non-fibrous 0MS sample. Conversely, the lowest UPV value was obtained from the 1.5MS sample, which contained the highest amount of fiber. This indicates that fiber negatively affected the fibrous geopolymer samples exposed to acid. Compared to the 1.5MS sample not exposed to acid, the UPV values of the 1.5MS samples exposed to acid for 14 and 28 days decreased by 0.36% and 0.96%, respectively. The reason for the high UPV value of the acid-exposed 0MS sample is that the metakaolin used as a binder material is resistant to acid effects, forming a chemically denser aluminosilicate gel in the matrix. Furthermore, the reason for the superiority of the UPV value of 0MS compared to other fiber-containing samples is that the fibers increase voids and create heterogeneity in the matrix [54]. Another reason is that increasing the fiber content in the geopolymer mortar affects workability and weakens the bridging property of the fiber [55].
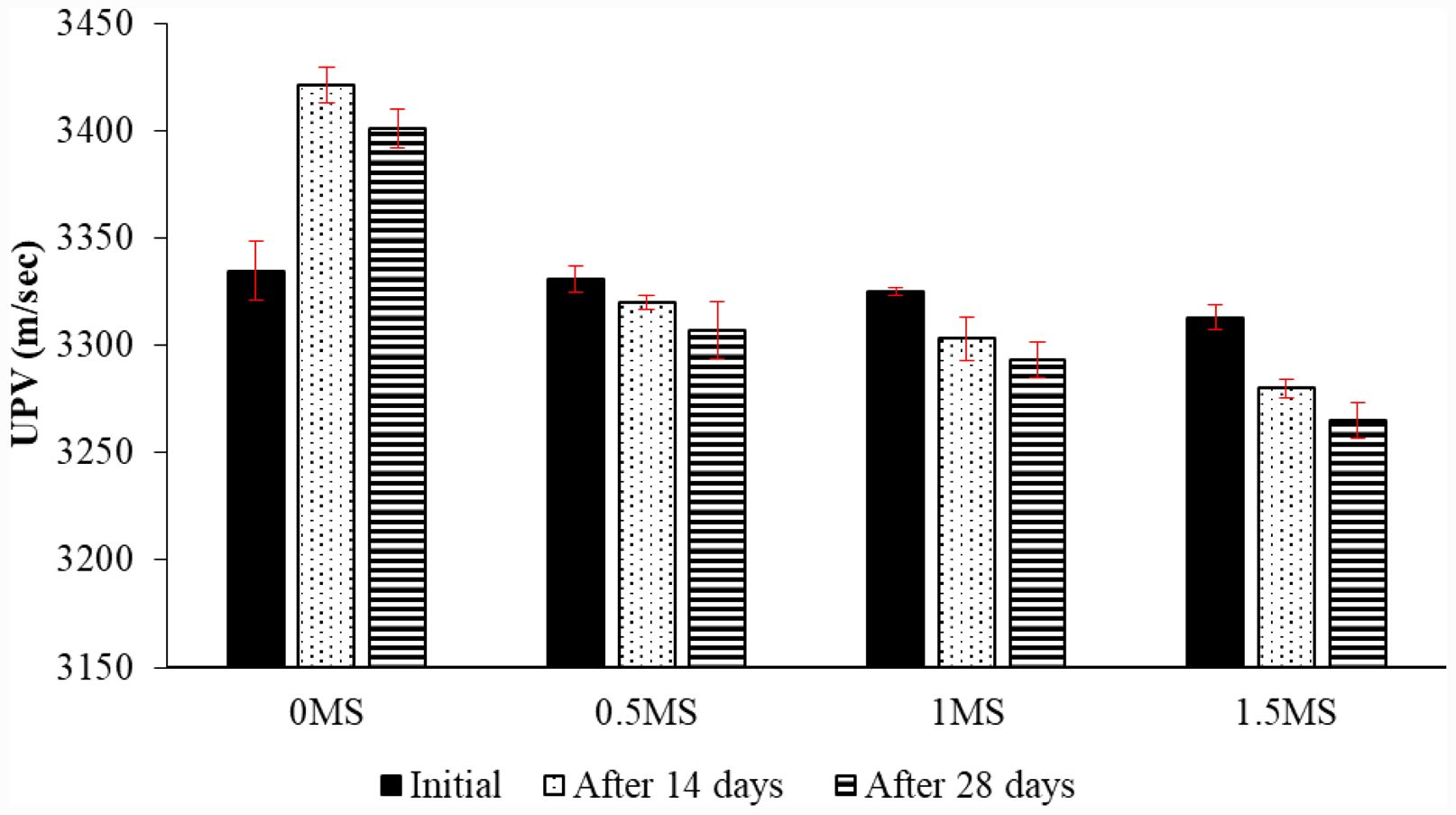
UPV of geopolymer mortars after sulfuric acid freeze-thaw effect.
Before and after 14 and 28 days of freeze-thaw cycles, the UPV for geopolymer mortar specimens with and without fiber is shown in Fig. 16. During 28 days of freeze-thaw cycles, the geopolymer specimens achieved their maximum UPV value of 0MS. Samples containing 1.5MS showed the lowest value after 28 days of freeze-thaw effect. Samples containing 1.5MS had a 0.99% drop in UPV value after 14 days and a 1.44% drop after 28 days compared to values before freeze-thaw cycles, respectively. Since metakaolin produces a denser and more chemically stable aluminosilicate gel, it is believed that this is the reason why geopolymer specimens without fiber had higher UPV values than samples with other fiber ratios. The study also documented a decrease in UPV corresponding to increased fiber content on days 14 and 28 due to the freeze-thaw effect. Microcracks form in the geopolymer matrix as a result of the fibers, which is associated with a slowing of the UPV [56].
Mass changes
Mass changes test of geopolymer specimens exposed to sulfuric acid
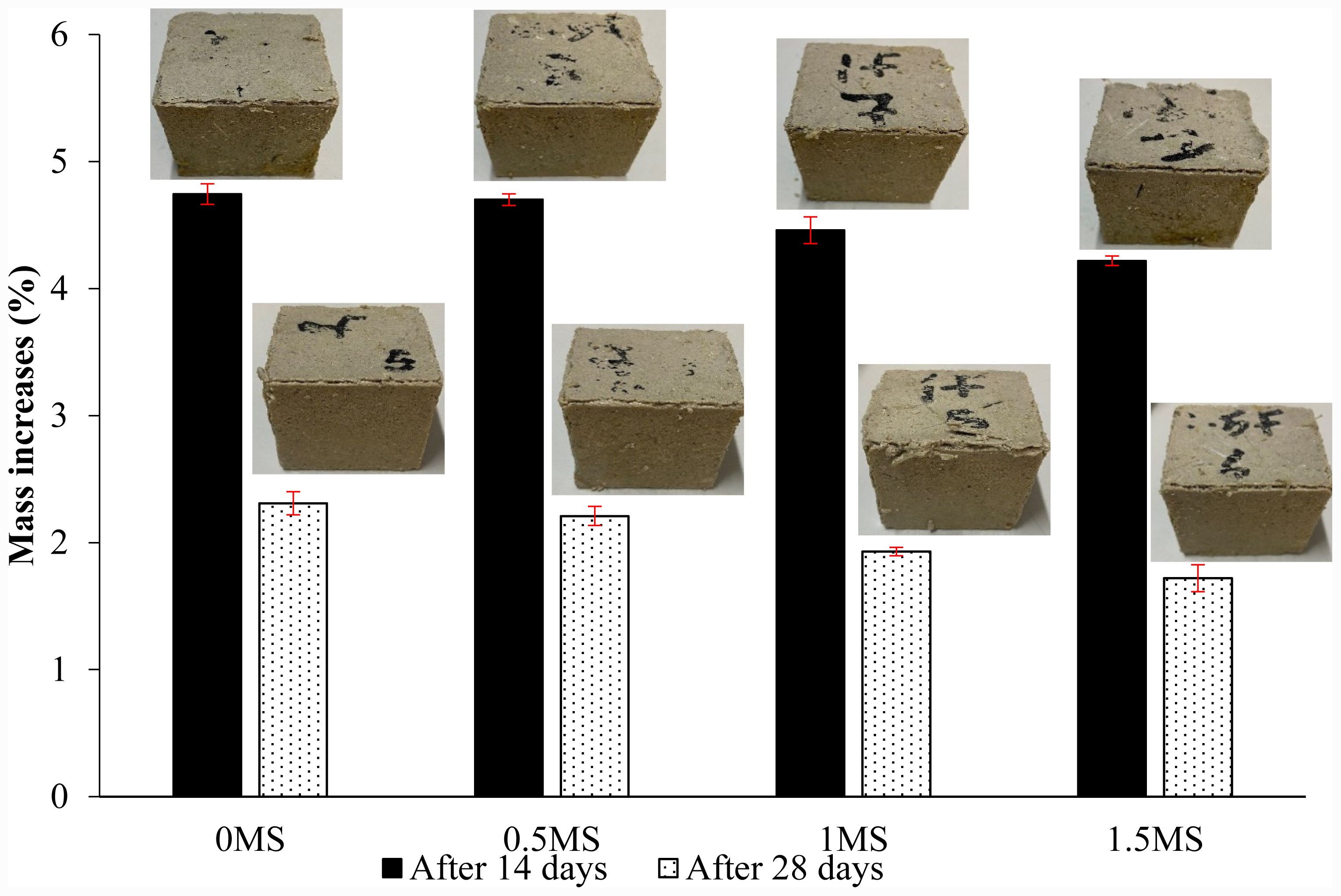
A mass change test was conducted on geopolymer mortar specimens with and without fiber composed of slag and metakaolin, and exposed to 5% sulfuric acid and freeze-thaw cycles to evaluate the stability of the geopolymer in harsh environments. Fig. 17 shows the results of the mass change test on samples exposed to sulfuric acid for 14 and 28 days. First, there was an increase in mass due to liquid absorption in all samples. In addition, there were different increases due to the chemical reactions in these samples [57]. The mass increases percentages for the 0MS, 0.5MS, 1MS, and 1.5MS samples were 4.74%, 4.70%, 4.46%, and 4.22% after 14 days of sulfuric acid exposure, respectively. The incorporation of fibers reduced mass gain in geopolymer specimens subjected to chemically demanding conditions, since these settings compromise the fiber–matrix interface, hence reducing connection and ingress routes. Thus, this degradation restricted the further infiltration of aggressive agents, thereby inhibiting subsequent mass growth [57]. Furthermore, the mass increases of the 0MS, 0.5MS, 1MS, and 1.5MS samples decreased to 2.31%, 2.21%, 1.93%, and 1.72%, respectively, after 28 days. The first mass rise in geopolymer specimens following 14 days of acid exposure is mostly due to the infiltration and retention of acidic solution inside the pore structure. Prolonged exposure of up to 28 days leads to the increasing leaching of alkali ions and the dissolution of aluminosilicate gel phases, resulting in surface degradation, material loss, and a subsequent decrease in the previously acquired mass [58].
Mass changes test of geopolymer specimens subjected to freeze-thaw cycle
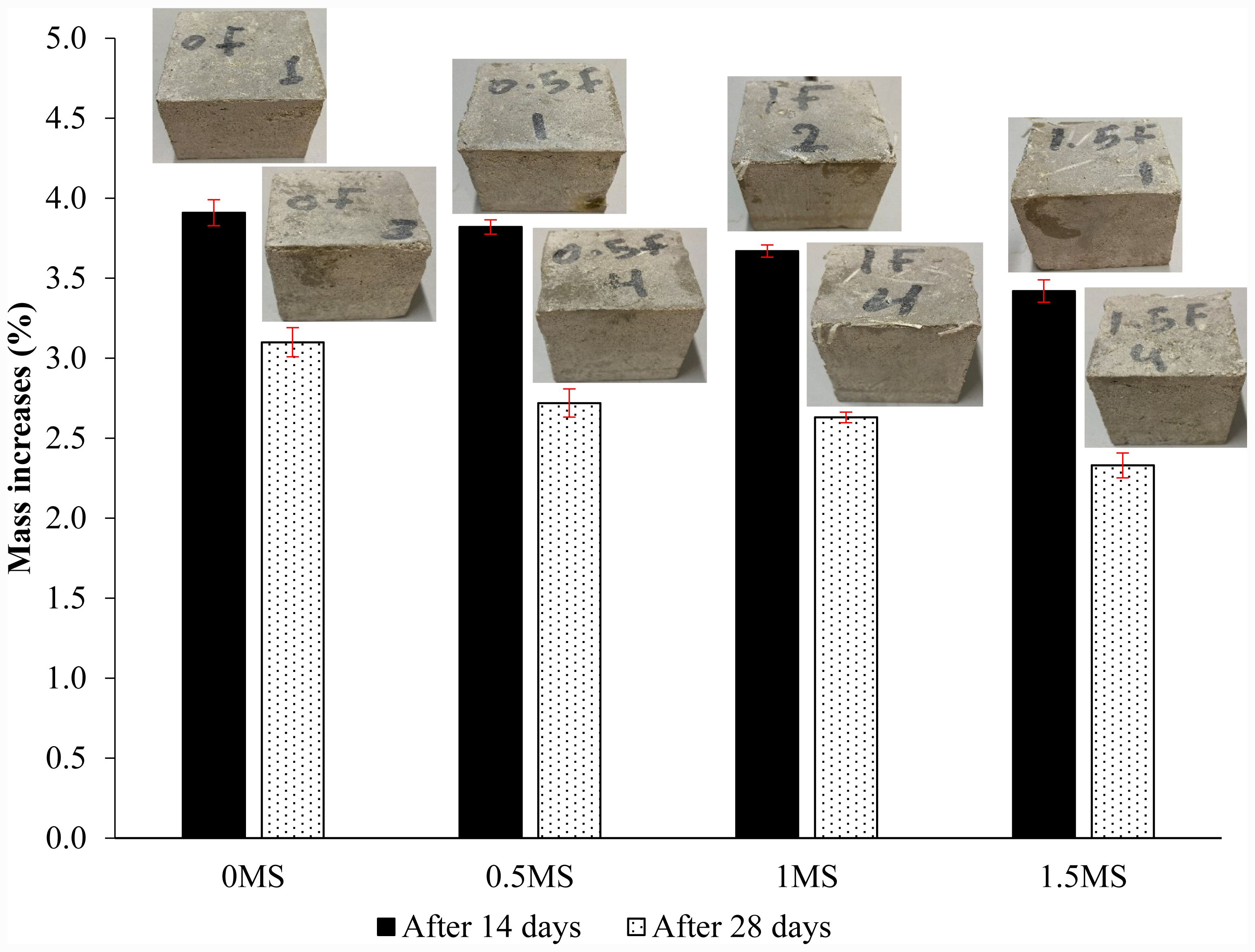
A mass change test was performed to assess the resilience of fiber-reinforced geopolymer mortar specimens that went through freezing and thawing in seawater. The outcomes of the mass change test on samples tested by cycles of freezing and thawing in seawater for 14 and 28 days are illustrated in Fig. 18. Initially, there was a mass augmentation attributable to liquid absorption in all samples. The mass of geopolymer specimens may increase following freeze-thaw exposure in seawater due to the infiltration of dissolved salts and ions into the pore structure during thawing. Repeated freezing cycles result in the precipitation and crystallization of these salts within the pores and microcracks, culminating in additional solid deposits and a perceived increase in mass [59]. The percentage increases in mass for the 0MS, 0.5MS, 1MS, and 1.5MS samples after 14 days of exposure to sulfuric acid were 3.91%, 3.82%, 3.67%, and 3.42%, respectively. Moreover, the mass increments of the 0MS, 0.5MS, 1MS, and 1.5MS samples decreased to 3.10%, 2.72%, 2.63%, and 2.33%, respectively, after a 28-day period. Prolonged exposure for up to 28 days to repeated freeze-thaw cycles results in gradual microcracking, scaling, and surface material loss, counteracting earlier water absorption and resulting in a decrease in previously acquired mass [60].
Microstructural analysis
0MS samples were selected before and after 28 days of seawater freeze-thaw treatment and after 28 days of sulfuric acid exposure. Fig. 19 shows scanning electron microscopy (SEM) examination of the samples before and after acid exposure and seawater freeze-thaw effects. Fig. 19(a) shows the 0MS sample, which exhibited a relatively compact and cohesive structure before acid exposure. This is because this sample contains no fibers and its internal structure consists of metakaolin and slag. The reactive aluminosilicate properties of metakaolin form the network of geopolymer gel, demonstrating the high strength of the sample under normal conditions [61]. Fig. 14(b) shows that after 28 days of freeze-thaw cycling in water, the 0MS sample exhibited microcracks extending throughout the open-pore matrix due to freezing pressure, but maintained its high density thanks to the presence of metakaolin [62]. Fig. 14(c) shows that after 28 days of acid treatment, the 0MS sample contained needle-shaped calcium sulfate crystals. Studies have shown that metakaolin resists acids [63].
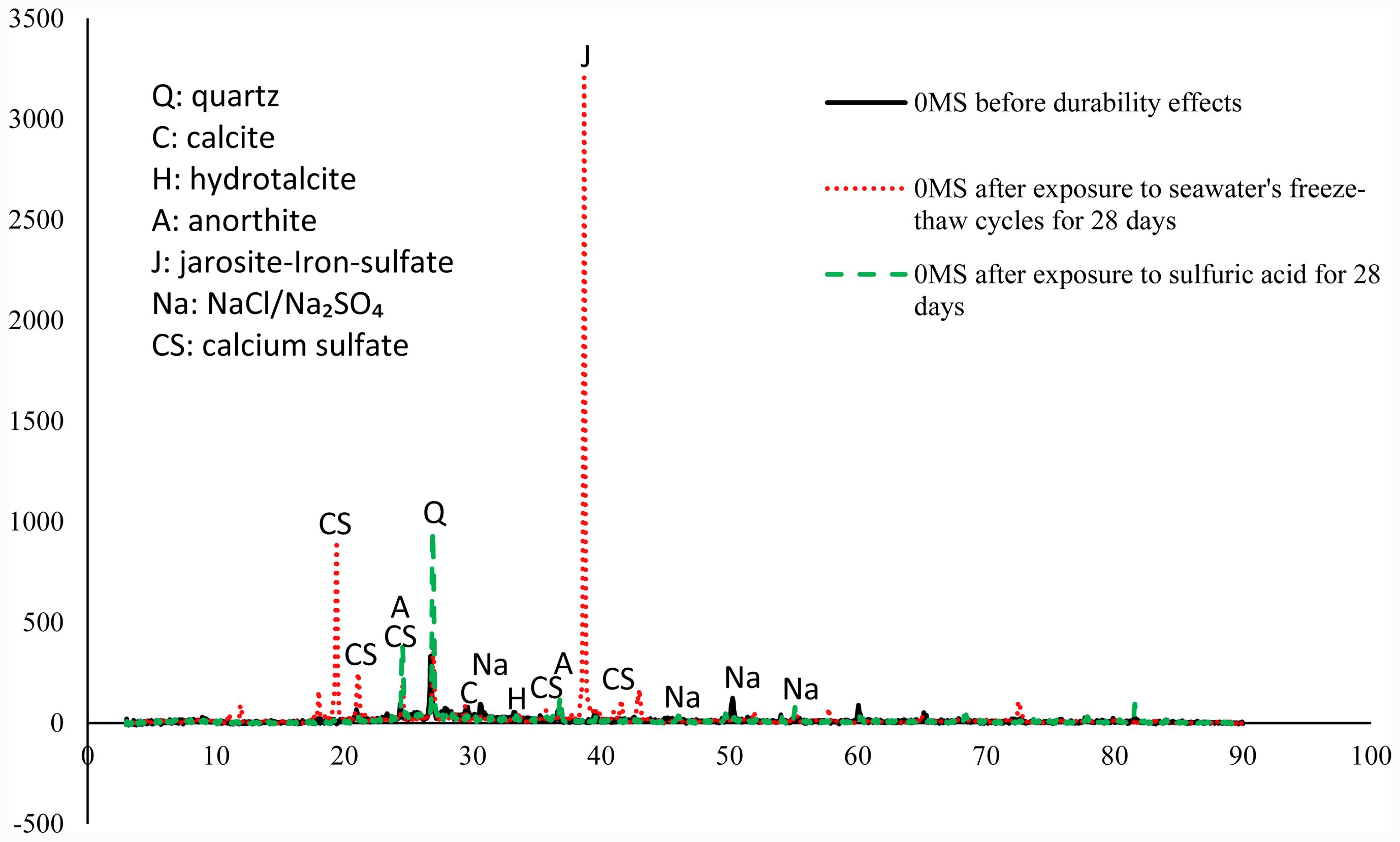
The XRD examination of 0MS samples before and after durability effects, shown in Fig. 20, was performed to detect crystalline phase alterations and evaluate the chemical stability of the geopolymer matrix under varying exposure circumstances [64]. This examination offers more data to reinforce the mechanical and microstructural findings. The enduring wide hump between 20–40° substantiates the existence of N-A-S-H/C-A-S-H gel, while the ~26.6–26.9° quartz peak remained invariant, underscoring the resilience of quartz from MK/sand to both chemical and freeze-thaw circumstances. Exposure to H2SO4 reveals new and enhanced reflections in the ~19–25° and ~36–41° zones, signifying the crystallization of calcium sulfates (gypsum and anhydrite), alongside a notably strong ~38.7° peak associated with jarosite-type iron sulfates resulting from slag-based Fe, indicating significant chemical degradation and calcium leaching. Concurrently, the diminishing intensity of the ~29.4° calcite peak aligns with decalcification and carbonate dissolution [65]. In contrast, samples exposed to seawater freeze–thaw cycles exhibit diminished peaks at approximately 31.7°, 45.5°, 50.0°, and 55.1°, indicating minor deposits of NaCl/Na2SO4. The continued presence of hydrotalcite-like H reflections (around 34.4–36.5°) signifies phase stability and a buffering capacity against sulfate degradation. The decrease in calcite intensity suggests partial leaching; however, the overall changes are less significant than those observed in acidic conditions, indicating that damage under freeze–thaw conditions is primarily physical (micro-cracking and increased porosity) rather than a chemical alteration of the matrix. Significantly, these XRD-based findings align well with the compressive strength findings, which also indicate more degradation with acid exposure than under freeze-thaw cycling.
|
Fig. 8 Mini-slump flow values of synthetic macro-fiber-based geopolymer mortar samples. |
|
Fig. 9 Amount of water absorbed per square meter of geopolymer samples with different synthetic macro fiber content. |
|
Fig. 10 Capillary water absorption coefficient of geopolymer mortars with different synthetic macro fiber content. |
|
Fig. 11 Compressive strength test of geopolymer mortar with various proportions of fibers and exposed to sulfuric acid. |
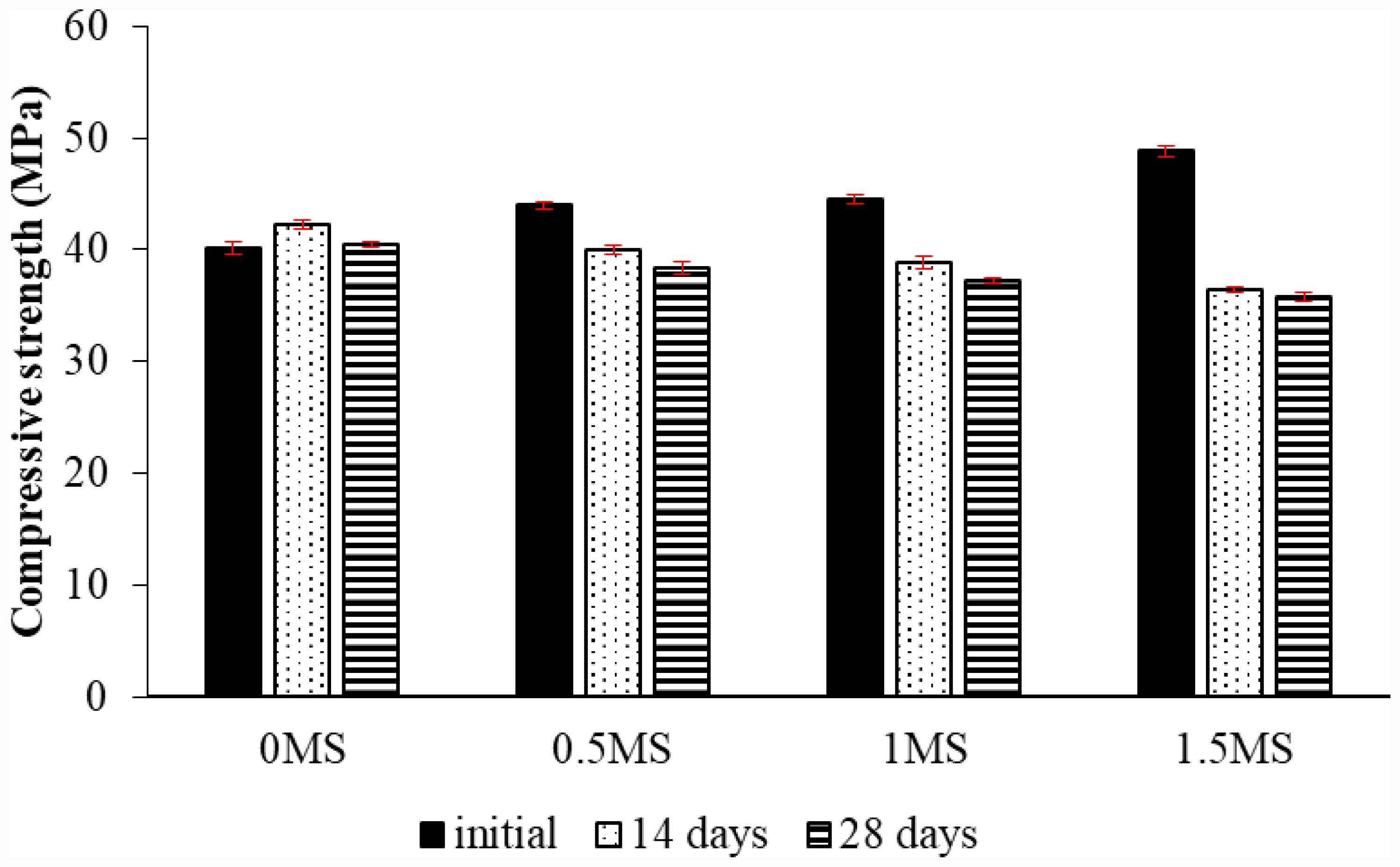
|
Fig. 12 Compressive strength testing of geopolymer mortar with various proportions of fibers, and subjected to freeze-thaw cycles. |
|
Fig. 13 Flexural strength test of geopolymer mortar with various proportions of fibers and exposed to sulfuric acid. |
|
Fig. 14 Flexural strength testing of geopolymer mortar with various proportions of fibers, and subjected to freeze-thaw cycles. |
|
Fig. 15 UPV of geopolymer specimens with and without fiber before and after acid effect. |
|
Fig. 16 UPV readings of geopolymer samples reinforced with various ratios of fiber before and after exposure to freeze-thaw cycles. |
|
Fig. 17 Mass change test data for synthetic macro-fibers -reinforced geopolymer specimens exposed to sulfuric acid. |
|
Fig. 18 Mass change test data for fiber-reinforced geopolymers tested by cycles of freezing and thawing in seawater. |

|
Fig. 19 SEM analysis of (a) 0MS before durability effects, (b) 0MS following exposure to seawater's freeze-thaw cycles for 28 days, and (c) 0MS following submergence in sulfuric acid for 28 days. |
|
Fig. 20 XRD analysis of the 0MS sample before durability effects, after exposure to seawater's freeze-thaw cycles for 28 days, and after exposure to sulfuric acid for 28 days. |
Several important conclusions about the effect of fiber inclusion, acid exposure, and freeze-thaw cycles in seawater on geopolymer mortar performance might be made from the experimental results:
Flow testing indicated that geopolymer samples containing fibers exhibited decreased flow values. Increasing the fiber content resulted in decreased flow properties. This indicates that the fibers impede the movement of the geopolymer mortar, increasing internal friction and reducing workability. Porosity and water absorption were also reduced for samples containing a high fiber content.
Compressive and flexural strength results showed a similar trend for samples exposed to sulfuric acid, where structural integrity deteriorated over time. In addition, a smaller decrease in strength was observed for samples during freeze-thaw cycles. This loss in strength was not as significant as in acidic environments. Acidic environments accelerated crack formation, while crack development was more limited during freeze-thaw cycles. However, the results showed that the sample with 1.5% fiber had the highest compressive strength and flexural strength, compared to the fiber-free sample prior to durability exposures.
Ultrasonic pulse velocity results also supported these findings, with samples exposed to acid showing a significant decrease in test values, while the decrease in test values during freeze-thaw cycles was more limited. The internal structure of the samples was found to be less damaged compared to those exposed to acidic environments. An increase in the mass of samples exposed to acidic environments was observed due to liquid absorption in the first few days, then a decrease due to chemical deterioration at 28 days. This was similar for samples exposed to freeze-thaw cycles, but at 28 days, the mass decreased due to microcracks.
It is concluded that the damage incurred in fiber-reinforced geopolymer samples exposed to acidic environments is larger, more pronounced, and more severe than in samples exposed to freeze-thaw cycles. This demonstrates their high resistance to physical impacts and low resistance to chemical and acidic effects.
The researchers of this work extend their appreciation to the employees who work in the Istanbul Aydın University-Civil Engineering Laboratory.
- 1. I. Bashir, K. Kapoor, and H. Sood, Int. J. Latest Res. Sci. Technol. 6[3] (2017) 33-36.
- 2. A. Fernández-Jiménez, M. Monzó, M. Vicent, A. Barba, and A. Palomo, Micropor Mesopor Mat. 108 (2008) 41-49.
-

- 3. D. Yan, S. Chen, Q. Zeng, S. Xu, and H. Li, Mater. Des. 95 (2016) 306-318.
-

- 4. V. Živica, S. Balkovic, and M. Drabik, Constr. Build. Mater. 25[5] (2011) 2206-2213.
-

- 5. Y.-C. Ding, Y.-S. Fang, and T.-W. Cheng, Mater. Struct. 49 (2016) 1883-1891.
-

- 6. X. Huang, T. Huang, S. Li, F. Muhammad, G. Xu, Z. Zhao, L. Yu, Y. Yan, D. Li, and B. Jiao, Ceram. Int. 42[8] (2016) 9538-9549.
-

- 7. C. Jithendra, V.N. Dalawai, and S. Elavenil, Mater. Today Proc. 51 (2022) 1580-1584.
-

- 8. B. Kim and S. Lee, J. Korean Ceram. Soc. 57 (2020) 368-377.
-

- 9. J.B. Aguiar, A. Camões, and P.M. Moreira, J. Adv. Concr. Technol. 6[1] (2008) 243-250.
-

- 10. W. Tahri, Z. Abdollahnejad, J. Mendes, F. Pacheco-Torgal, and J.B. Aguiar, Eur. J. Environ. Civ. Eng. 21[5] (2017) 555-571.
-

- 11. J. Davidovits, Proc. First Int. Conf. Alkaline Cem. Concr. 1 (1994) 131-149.
- 12. Z.M.K. Al-Mayali and M. Ziada, Res. Eng. Struct. Mater. 11[4] (2025) 1749-1765.
-

- 13. M. Ziada and Z.M.K. Al-Mayali, J. Sustain. Constr. Mater. Technol. 10[2] (2025) 248-262.
-

- 14. X.Y. Zhuang, L. Chen, S. Komarneni, C.H. Zhou, D.S. Tong, H.M. Yang, W.H. Yu, and H. Wang, J. Clean. Prod. 125 (2016) 253-267.
-

- 15. T.C. Powers and R.A. Helmuth, Highw. Res. Board Proc. (1953) 131-149.
- 16. K. Ebrahimi, M.J. Daiezadeh, M. Zakertabrizi, F. Zahmatkesh, and A.H. Korayem, Constr. Build. Mater. 186 (2018) 1105-1113.
-

- 17. Y. Fu, L. Cai, and W. Yonggen, Constr. Build. Mater. 25[7] (2011) 3144-3148.
-

- 18. P. Sun and H.-C. Wu, Fuel 111 (2013) 740-745.
-

- 19. R. Brooks, M. Bahadory, F. Tovia, and H. Rostami, Int. J. Sustain. Eng. 3[3] (2010) 211-218.
-

- 20. Y. Yuan, R. Zhao, R. Li, Y. Wang, Z. Cheng, F. Li, and Z.J. Ma, Constr. Build. Mater. 250 (2020) 118831.
-

- 21. Y. Aygörmez, O. Canpolat, M.M. Al-Mashhadani, and M. Uysal, Constr. Build. Mater. 235 (2020) 117502.
-

- 22. M. Ahmed, Q.Q. Liang, M.N. Sheikh, and M.N.S. Hadi, Eng. Struct. 283 (2023) 115882.
-

- 23. Y. Yang, Q. Zhou, Y. Deng, and J. Lin, Cem. Concr. Compos. 128 (2022) 104422.
-

- 24. M. Ziada, H. Tanyildizi, A. Coskun, and M. Seloglu, Struct. Concr. 26[3] (2025) 3096-3107.
-

- 25. F.U.A. Shaikh, Constr. Build. Mater. 43 (2013) 37-49.
-

- 26. M. Ziada, S. Erdem, R. Alonso González-Lezcano, Y. Tammam, and İ. Unkar, Eng. Sci. Technol. Int. J. 46 (2023) 101501.
-

- 27. A.R.G. de Azevedo, A.S.A. Cruz, M.T. Marvila, L.B. de Oliveira, S.N. Monteiro, C.M.F. Vieira, R. Fediuk, R. Timokhin, N. Vatin, and M. Daironas, Polymers (Basel) 13[15] (2021) 2493.
-

- 28. W. Li and J. Xu, Mater. Sci. Eng. A 505 (2009) 178-186.
-

- 29. D.P. Dias and C. Thaumaturgo, Cem. Concr. Compos. 27[1] (2005) 49-54.
-

- 30. N. Ranjbar, S. Talebian, M. Mehrali, C. Kuenzel, H.S.C. Metselaar, and M.Z. Jumaat, Compos. Sci. Technol. 122 (2016) 73-81.
-

- 31. P. Duxson, S.W. Mallicoat, G.C. Lukey, W.M. Kriven, and J.S.J. van Deventer, Colloids Surf. A Physicochem. Eng. Asp. 292[1] (2007) 8-20.
-

- 32. K. Zhang, H. Xu, X. Kong, C. Zhang, H. Lu, and D. He, J. Build. Eng. 76 (2023) 107164.
-

- 33. ASTM C109/C109M-02 ASTM Int. (2020).
- 34. ASTM C 348 – 21, Norma ASTM Int. (2021).
- 35. ASTM C1585, ASTM Int. (2020).
- 36. ASTM C597-09, ASTM Int. (2016).
- 37. B. Nematollahi, J. Sanjayan, and F.U.A. Shaikh, J. Mater. Civ. Eng. 27[10] (2015) 04015001.
-

- 38. N.S. Deepa Raj, R. Abraham, N. Ganesan, and D. Sasi, Proc. Int. Conf. Innov. Civ. Eng. (2013) 75-80.
- 39. V.S. Sujitha, S. Raja, M.A. Rusho, and S. Yishak, Case Stud. Constr. Mater. 22 (2025) e04669.
-

- 40. B.B. Jindal, T. Alomayri, A. Hasan, and C.R. Kaze, Environ. Sci. Pollut. Res. 30 (2023) 25299-25324.
-

- 41. M. Ohno and V.C. Li, Indian Concr. J. 93[12] (2019) 47-53.
- 42. M. Vafaei, A. Allahverdi, P. Dong, N. Bassim, and M. Mahinroosta, J. Build. Eng. 34 (2021) 102066.
-

- 43. J. Kwasny, T.A. Aiken, M.N. Soutsos, J.A. McIntosh, and D.J. Cleland, Constr. Build. Mater. 166 (2018) 537-553.
-

- 44. R. Çetintaş and S. Soyer-Uzun, J. Build. Eng. 20 (2018) 130-136.
-

- 45. T. Jiang, Z. Liu, X. Tian, J. Wu, and L. Wang, Constr. Build. Mater. 412 (2024) 134760.
-

- 46. M. Ziada, H. Tanyildizi, and M. Uysal, Constr. Build. Mater. 414 (2024) 135047.
-

- 47. M. Jin, Z. Wang, F. Lian, and P. Zhao, Constr. Build. Mater. 265 (2020) 120730.
-

- 48. D. Niu, L. Su, Y. Luo, D. Huang, and D. Luo, Constr. Build. Mater. 237 (2020) 117628.
-

- 49. S. Pang, X. Zhang, B. Lei, H. Fan, J. Liu, P. Ju, and Y. Gao, Constr. Build. Mater. 452 (2024) 138943.
-

- 50. H.G. Alcan, B. Bayrak, A. Öz, O. Çelebi, G. Kaplan, and A.C. Aydın, Constr. Build. Mater. 414 (2024) 135059.
-

- 51. H.J. Zhuang, H.Y. Zhang, and H. Xu, Procedia Eng. 210 (2017) 126-131.
-

- 52. J. Cao, H. Peng, B. Zhang, Z. Yang, H. Pan, J. Chen, and X. Zhao, Constr. Build. Mater. 493 (2025) 143129.
-

- 53. A. Rajaee, N. Talebi, and S. Abrishami, Case Stud. Constr. Mater. 22 (2025) e04439.
-

- 54. B.T. Gebretsadik, M.S. Thesis, University of Nevada (2013).
-

- 55. S. Hedjazi and D. Castillo, Heliyon 6[3] (2020) e03646.
-

- 56. N. Ali, O. Canpolat, Y. Aygörmez, and M.M. Al-Mashhadani, Constr. Build. Mater. 251 (2020) 118976.
-

- 57. M.G. Sá Ribeiro, P.F. Keane, M.R. Sardela, W.M. Kriven, and R.A. Sá Ribeiro, Constr. Build. Mater. 302 (2021) 124194.
-

- 58. M.A.M. Ariffin, M.A.R. Bhutta, M.W. Hussin, M. Mohd Tahir, and N. Aziah, Constr. Build. Mater. 43 (2013) 80-86.
-

- 59. M.H.H. Rahat and A.S. Brand, Constr. Build. Mater. 489 (2025) 142363.
-

- 60. S. Pilehvar, A.M. Szczotok, J.F. Rodríguez, L. Valentini, M. Lanzón, R. Pamies, and A.-L. Kjøniksen, Constr. Build. Mater. 200 (2019) 94-103.
-

- 61. J. Yang, D. Li, and Y. Fang, Constr. Build. Mater. 167 (2018) 65-72.
-

- 62. S.M.S. Kazmi, M.J. Munir, Y.-F. Wu, I. Patnaikuni, Y. Zhou, and F. Xing, Cold Reg. Sci. Technol. 178 (2020) 103126.
-

- 63. Y. Aygörmez and O. Canpolat, Rev. Construcción 20[2] (2021) 291-307.
-

- 64. I. Garcia-Lodeiro, A. Palomo, A. Fernández-Jiménez, and D.E. Macphee, Cem. Concr. Res. 41[9] (2011) 923-931.
-

- 65. M. Kobayashi, K. Takahashi, and Y. Kawabata, Materials 16[15] (2023) 5278.
-

 This Article
This Article
-
2026; 27(1): 150-162
Published on Feb 28, 2026
- 10.36410/jcpr.2026.27.1.150
- Received on Dec 25, 2025
- Revised on Jan 5, 2026
- Accepted on Jan 9, 2026
 Services
Services
- Abstract
introduction
substances and methodologies
results
conclusion
- Acknowledgements
- References
- Full Text PDF
Shared
 Correspondence to
Correspondence to
- Mahmoud Ziada
-
Department of Civil Engineering, Istanbul Aydin University, Istanbul, Türkiye
Tel : +90 444 14 28 / 23101 Fax: +90 212 425 57 59 - E-mail: mahmoudm.s.ziada@aydin.edu.tr






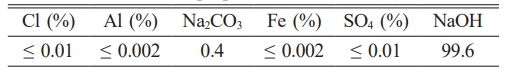

 Copyright 2019 International Orgranization for Ceramic Processing. All rights reserved.
Copyright 2019 International Orgranization for Ceramic Processing. All rights reserved.